Animation Cathode Ray Experiment
6 The Effect of an Electric Field on.
Animation cathode ray experiment. Scientists in the early nineteenth century were aware of electricity and the effect of electric potential. Cathode ray SmallCircle alpha particles proton beam neutron beam Who performed this experiment?. The magnitude of the electric current flowing.
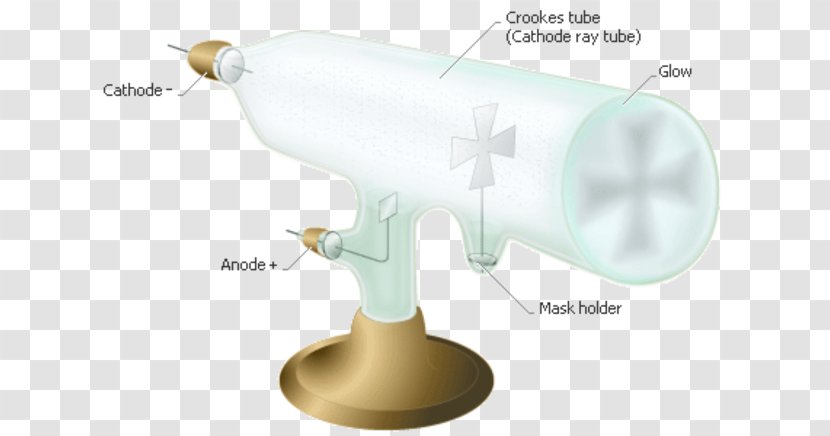
The pair, in turn, was connected to an electrometer, a device for catching and measuring electric charges. He did this using a cathode ray tube or CRT. Regions in a Crookes tube.
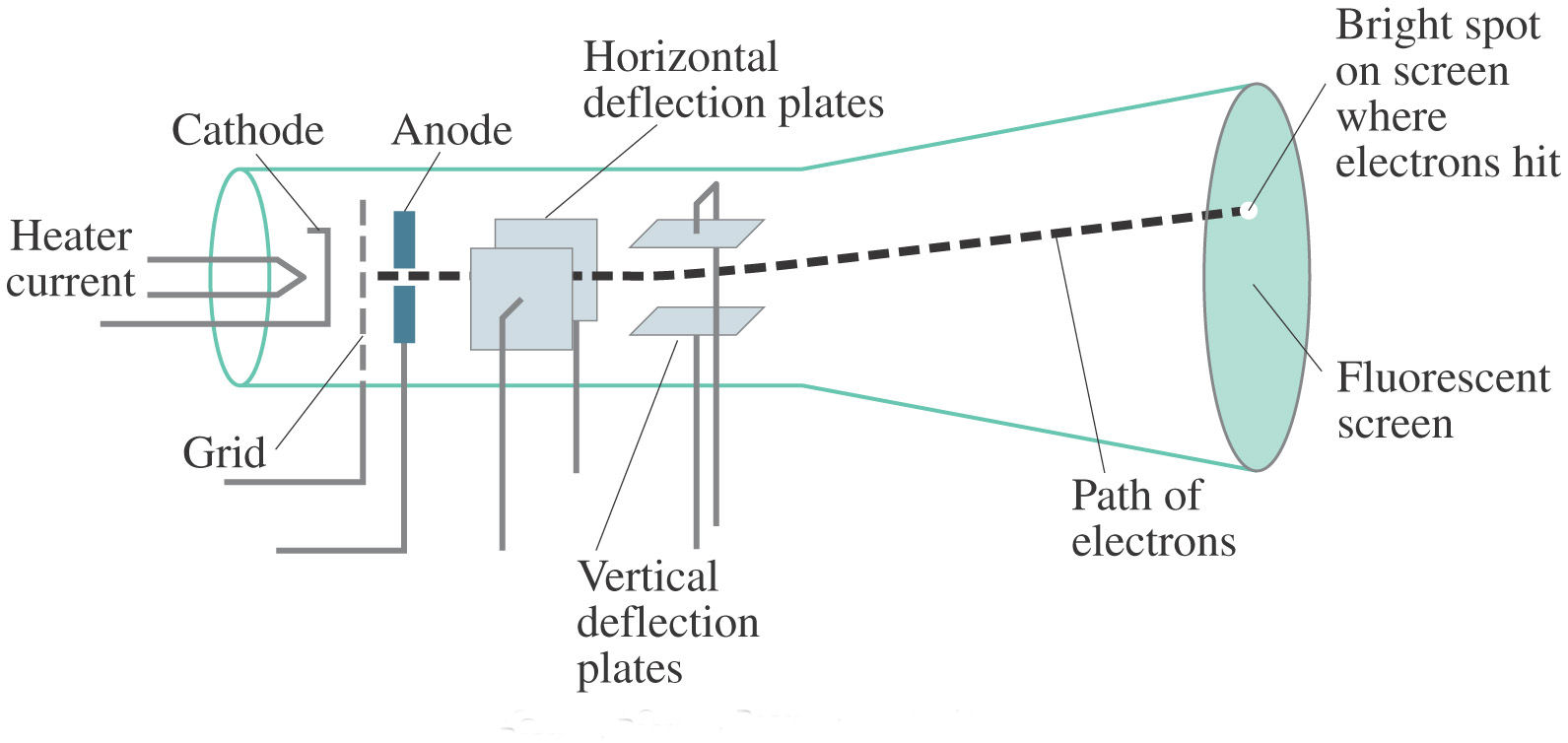
The experiment Cathode Ray Tube (CRT) conducted by J. Thomson showed that cathode rays were. The following block diagram shows the general purpose CRO contraction.The CRO recruit the cathode ray tube and acts as a heat of the oscilloscope.
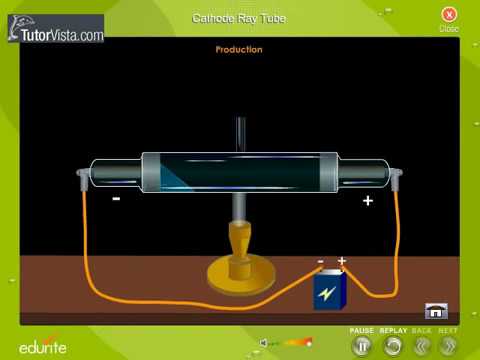
Experiments with cathode ray tubes and tried to use the results to determine the nature of cathode rays. Cathode Ray Tube Animation. Then, on operating the CRT, in the absence of.
Cathode Ray Experiment After Dalton's breakthrough of atomic theory, scientists tried to determine the masses of atoms from the fraction of elements in compounds. Rutherford gold foil experiment. Cathode Ray Tube The Cathode Ray Tube (CRT) is a vacuum tube containing an electron gun (a source of electrons) and a fluorescent screen, with internal or ex.
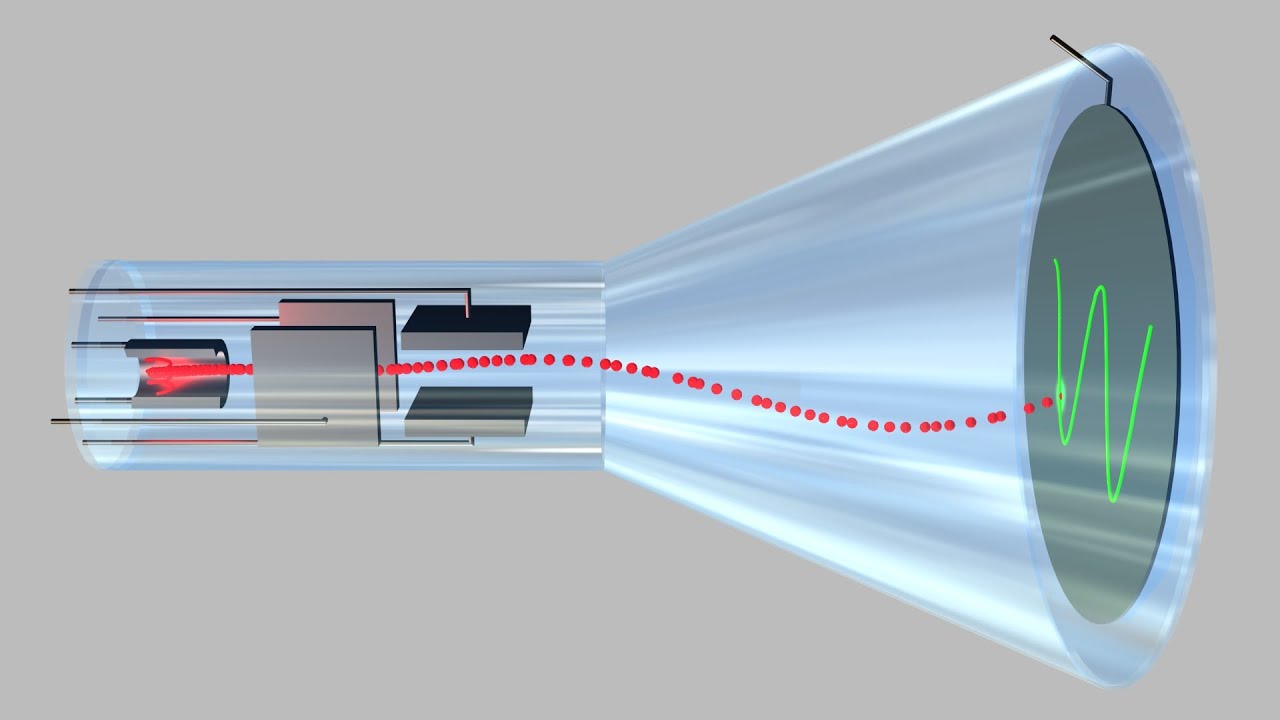
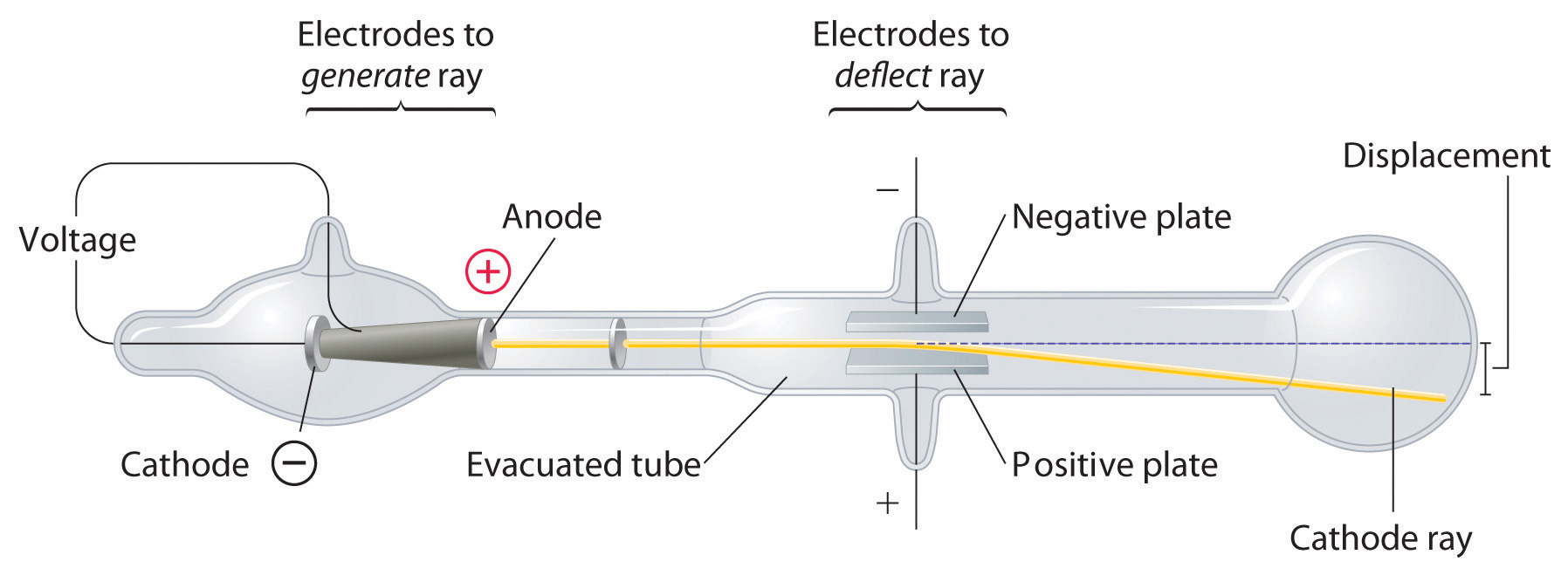
Thomson is the first individual to succeed in deflecting the cathode ray with an electrical field. Turn the magnet on and off to see how the beam changes. If an evacuated glass tube is equipped with two electrodes and a voltage is applied, glass behind the positive electrode is observed to glow, due to electrons emitted from the cathode.
The history of atomic chemistry. You can create videos from my animations and place them, for example on youtube. The term Crookes tube is also used for the first generation, cold cathode X-ray tubes, which evolved from the experimental Crookes tubes and were used until about 19.
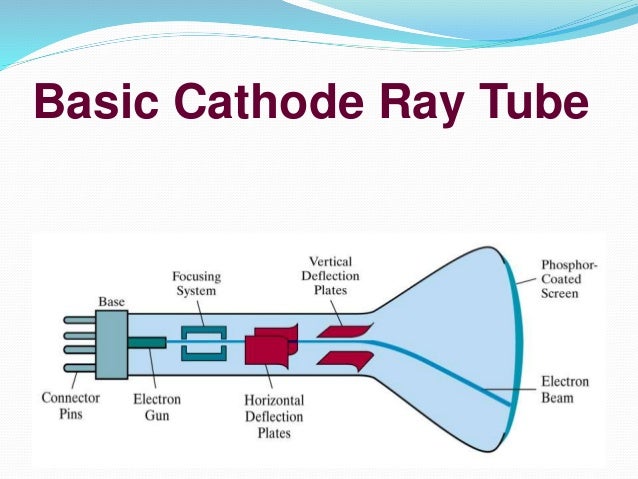
Thomson (1856 – 1940) J. A CRO is an electronic device with a CRT as its main component and other associated circuits consisting of a power supply unit, a sawtooth-wave generator, horizon and vertical amplifiers. Discovery of the electron and nucleus.
Explain what is a cathode ray oscilloscope (CRO)?. Cathode rays focused on a hard target (anticathode) produce X-rays or focused on a small object in a vacuum generate very high temperatures (cathode-ray furnace). (Right-click here to download).
The apparatus of his experiment is called the cathode-ray tube (CRT). QUESTION 4 OF 5 A cathode ray tube is the device used in television sets. Thomson studied cathode ray tubes and came up with the idea that the particles in the cathode beams must be negative because they were repelled by negatively charged items (either the cathode or a negatively charged plate in the cathode ray tube) and attracted by positively charged items (either the anode or the positively charged plate in the cathode ray tube).
The picture below illustrates the operation of a Crookes tube in a schematic way. Flash Animations for Physics > Thomson's experiment. Scientists used special vacuum tubes, such as the Crookes tube and the cathode ray tube, to study this phenomenon.
Explain what is a cathode ray oscilloscope (CRO)?. $\endgroup$ – matt_black Dec 13 '18 at 14:05. The cathode ray oscilloscope is a vital piece of diagnostic lab equipment for observing and measuring electrical signals at frequencies ranging from dc to GHz Watch full Video.
Cathode Ray Oscilloscope Animation. Anode(+ve) and the cathode (-ve).The tube was filled with an inert gas.A perforated or porous cathode was used. If you then want to know either the charge or the mass of an electron, you need to have a way of measuring one or the other independently.
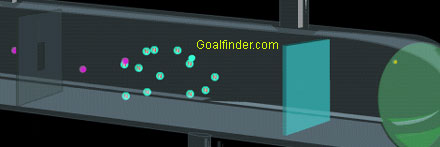
→These rays are also known as canal rays. Here is an animation of a famous scientific experiment. Thomson noted that the rays inside the tube were deflected, which was inferred as the presence of a negatively charged particle inside the vacuum tube that made such a phenomenon possible.
Google Classroom Facebook Twitter. Thomson took a cathode ray tube and at the place where the electron beam was supposed to strike, he positioned a pair of metal cylinders having slits on them. Discovery of electron took a long journey over hundreds of years from lightning to discharge tubes to cathode rays and finally discovery of electron by J.
The discovery of electrons in chemistry was substantiated by an electric discharge in cathode-ray tubes. Thomson, is one of the most well-known physical experiments that led to electron discovery. The diagrams help students understand the significance of each experiment.
Since an individual atom is so small, the mass of the atoms of one of the element was determined relative to the mass of the atoms of another element, based on a mass standard. In fact, if you are reading this on a non-flat panel desktop computer monitor, or have ever viewed a non-flat screen television. This paper describes the simulation of J.
Crookes also demonstrated that magnetic fields could deflect cathode rays. The major contribution of this work is the new approach for modelling this experiment, with a great deal of accuracy and precision, using the equations of physical laws to describe the motion of the electrons. Cathode ray tubes are not an outdated piece of scientific equipment;.
This paper describes how J is simulated. Electrical Q & A;. Thomson was not the only one working on cathode rays, but several other players like Julius Plücker, Johann Wilhelm Hittorf, William Crookes, Philipp Lenard had contributed or were busy studying it.
He built his cathode ray tube with a metal cylinder on the other end. A class activity sheet accompanies the cathode ray tube demonstrations and experiments. His first experiment was to build a cathode ray tube with a metal cylinder on the end.
It is a vacuum sealed tube with a cathode and anode on one side. Thomson's cathode ray experiment was a set of three experiments that assisted in discovering electrons. J Thomson, this 50 minute animation looks at every aspect of this discovery.
You can change the value of the electric field and play it with the magnetic field, of 2 mT, on or off. Niels Bohr J.J Thomson Ernest Ruthorford Robert Millikan What did this experiment demonstrate?. By Staff Writer Last Updated Apr 10, 3:47:37 AM ET.
E) of an electron. BUT the maltose cross provided a distinct well formed shadow on the end of the tube. S Bharadwaj Reddy March 4, 15 September 11, 16.
In addition, the experiment could describe characteristic properties, in essence, its affinity to positive charge, and its charge to mass ratio. 4) Although there was some speculation that the cathode rays were negatively charged, it is not shown to be true by experiment until 15, just two years before Thomson announces the electron. →This experiment helped in the discovery of the proton.
Chapter 9 - Models of Chemical Bonding Rutherford's Experiment 2 Ionic Versus Covalent Bonds:. Click on the portion of the diagram where the television screen would be located. Cathode ray, stream of electrons leaving the negative electrode (cathode) in a discharge tube containing a gas at low pressure, or electrons emitted by a heated filament in certain electron tubes.
Canal Ray experiment is the experiment performed by German scientist Eugen Goldsteinin 16 that led to the discovery of the proton. In 1879, Sir William Crookes demonstrated that cathode rays travel in straight lines by using the tube shown at the right. In the Thomson Cathode Ray Tube Experiment, it was discovered that you can use the deflection of an electron beam in an electric and magnetic field to measure the charge-to-mass ratio (q / m.
CRO is an electronic device that gives graphical representation of alternating quantities under. Educational atomic physics animation :. This is the currently selected item.
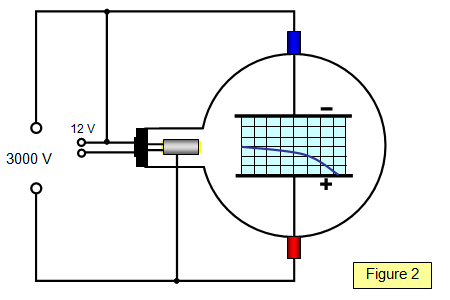
Your job is to measure the deflection of the beam under different conditions and then determine the ratio of the charge of the particles in the beam to the mass of the particles in the beam. Thomson's experiment with cathode rays (position is given in mm, 10 −3 m). Thomson’s First Cathode Ray Experiment.
How is CRO superior to ordinary measuring instruments?. A CRO is an electronic device with a CRT as its main component and other associated. Cathode Ray consist of negatively charged particles called electrons.
Chapter 8 - Electron Configuration and Chemical Periodicity Millikan Oil Drop Experiment Electron Configs Rutherford's Experiment 1:. Cathode ray oscilloscope experiment viva questions. A Cathode Ray Tube Zumdahl, Zumdahl, DeCoste, World of Chemistry 02, page 58 5 Cathode Ray Experiment.
Thomson's Cathode Ray Tube experiment. Electrons in cathode ray tubes behave far more like particles than waves and the relevant experiment was a major breakthrough in our understanding of matter well before quantum stuff became important. Cathode Ray Tube Emission Spectrum JJ Thomson's Experiment:.
Rutherford gold foil experiment. What is the principle of operation of cathode ray oscilloscope?. →Anode ray experiment was conducted by E Goldstein.
The animation illustrates the deflection of an electron in external fields which simulates J. It figured out a bunch of details about electrons and specially that the atom is not the smallest particle in matter. It is a glass tube from whichmost.
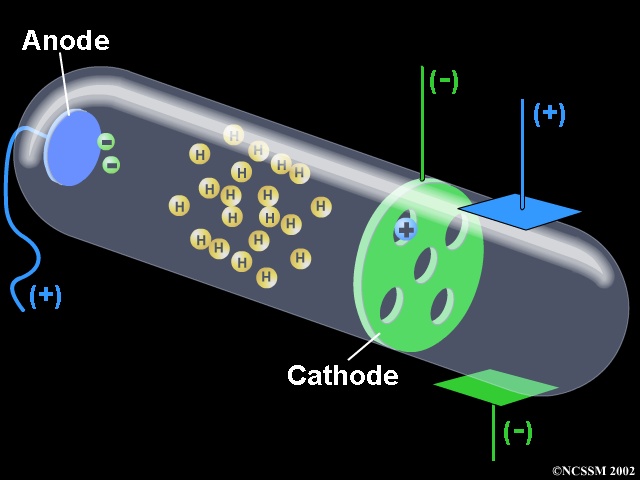
Apparatus Used A discharge tube was taken in which there were 2 electrodes i.e. The Maltose cross was placed midway in the cathode ray tube. Sim1 0,0,359,0 360,0,440,0 1 false true standard %CORRECT% The electrons are accelerated to the right and strike the television screen to the right of the anode.
Chapter 3 - Stoichiometry. They were first observed in 1869 by German physicist Julius Plücker and Johann Wilhelm Hittorf, and were named in 1876 by Eugen Goldstein Kathodenstrahlen, or cathode rays. Thomson’s Cathode Ray Experiment.
The rays bent towards the positive pole, indicating that they are negatively charged. This experiment was performed using a cathode ray tube (Crooke’s tube). The discovery of proton which happened after the discovery of electron further strengthened the structure of the atom.In the experiment, Goldstein applied high voltage across a discharge tube which had a perforated cathode.
Thomson's cathode ray experiment and Rutherford's gold foil experiment. Thompson, conducted the cathode ray tube experiment to prove that rays emitted from an electron gun are inseparable from the latent charge. How the deflection of of electrons occur in cathode ray tube?.
It can be used to determine the velocity of the electrons (when they are not deflected by the electric and magnetic fields) and the charge-to-mass ratio. Define cathode ray tube with help of a well labeled diagram?. Travelling in straight lines and and producing a shadow from radiated waves indicated a wave nature.
Developed from the earlier Geissler tube, the Crookes tube consists of a partially evacuated glass bulb of various shapes, with two metal. The activity sheet has embedded questions designed to promote small group discussions. Cathode Ray Oscilloscope Block Diagram of CRO.
The electric potential is a driving force that results in the flow of current through a substance due to the difference in concentration of charges at two ends of it. In 17, British physicist J. A portrait of J.
State whether the statement is true or false “A negative high voltage is applied to the filament to emit electrons” and justify your answer?. When a gas is subjected to a high potential (5000 to V) at low pressure, the glass wall of tube glows with fluorescent light. In an oscilloscope, the CRT produces the electron beam which is accelerated to a high velocity and brings to the focal point on a fluorescent screen.
This is the official Video of Cathode Ray Tube by sir JJ ThomsonA Cathode ray tube is the forerunner of the television tube. The waveform is generated in such a way that the amplitude of the signal is represented along Y-axis and the variation in the time is represented along X-axis. History of atomic structure.
The tube illustrated in the tutorial contains a negative electrode (Cathode) at one end and a positive electrode (Anode) at the other.A high voltage is transmitted to the cathode ray tube, inducing the cathode to emit electrons – essentially an electrical current. Thomson's cathode ray tube experiment. Thomson’s First CRT Experiment.
Using a cathode ray tube, Thomson was able to deflect cathode rays with an electrical field. Thomson had an inkling that the ‘rays’ emitted from the electron gun were inseparable from the latent charge, and decided to try and prove this by using a magnetic field. There is a side tube connected to a vacuum pump to reduce pressure.
The activity sheet has diagrams of each of the cathode ray tubes used in the demonstration. It consists of a glass tube connected to two metal electrodes at two ends. Cathode rays are streams of electrons observed in discharge tubes.
In 1878 William Crooks observed that a beam of light travels from cathode to the anode, when he lowered the pressure of the Crooks Tube with a gas to 1Pa. Thomson's Cathode Ray Tube Lab In this lab we will be looking at a model of the Thomson experiment using a Cathode Ray Tube. A Crookes tube (also Crookes–Hittorf tube) is an early experimental electrical discharge tube, with partial vacuum, invented by English physicist William Crookes and others around 1869-1875, in which cathode rays, streams of electrons, were discovered.
What was the cathode ray tube experiment conclusion?. Crookes tube - Maltese cross tube. The cathode ray oscilloscope is the instrument which generates the waveform of any electrical quantity.
For experiments with cathode ray tube we used an educational model readily available on eBay.

Cathode Ray Tube Experiment Jj Thomson Cathode Ray Tube Tecnologia
541 Cathode Ray Tube Of Oscilloscope Youtube

Learn Electron And It S Discovery In 3 Minutes
Animation Cathode Ray Experiment のギャラリー

Electromagnetic Deflection In A Cathode Ray Tube I Maglab

Atoms And Periodic Properties Ppt Download

Cathode Ray Tube Experiments Physicsopenlab

The Atomic Theory Of Matter Summary

Animations List Bauer

Atomic Discovery Early Models Of The Atom 400 B C Democritus Proposed The Existence Of Fundamental Particles Of Matter That Were Indivisible And Indestructible Ppt Download
Goalfinder Discovery Of Proton Animated Easy Science Technology Software Online Education Medical K12 Animation E Learning

Atomic Theory I Chemistry Visionlearning

Cathode Ray Tube Animated Explanation Youtube

What Are Cathode Ray Experiment And Anode Ray Experiment Chemistry Topperlearning Com Oxijwfuu
Tiger Ncssm Distance Education And Extended Programs

2 4 Early Experiments To Charachterize The Atom Apchemwins

History Of Atoms Storyboard By 52a16d5b

Discovery Of Electron Cathode Rays Discharge Tube Experiment Full Chemistry Animation Facts Youtube
J J Thomson Storyboard By Jegugael1
The Atom
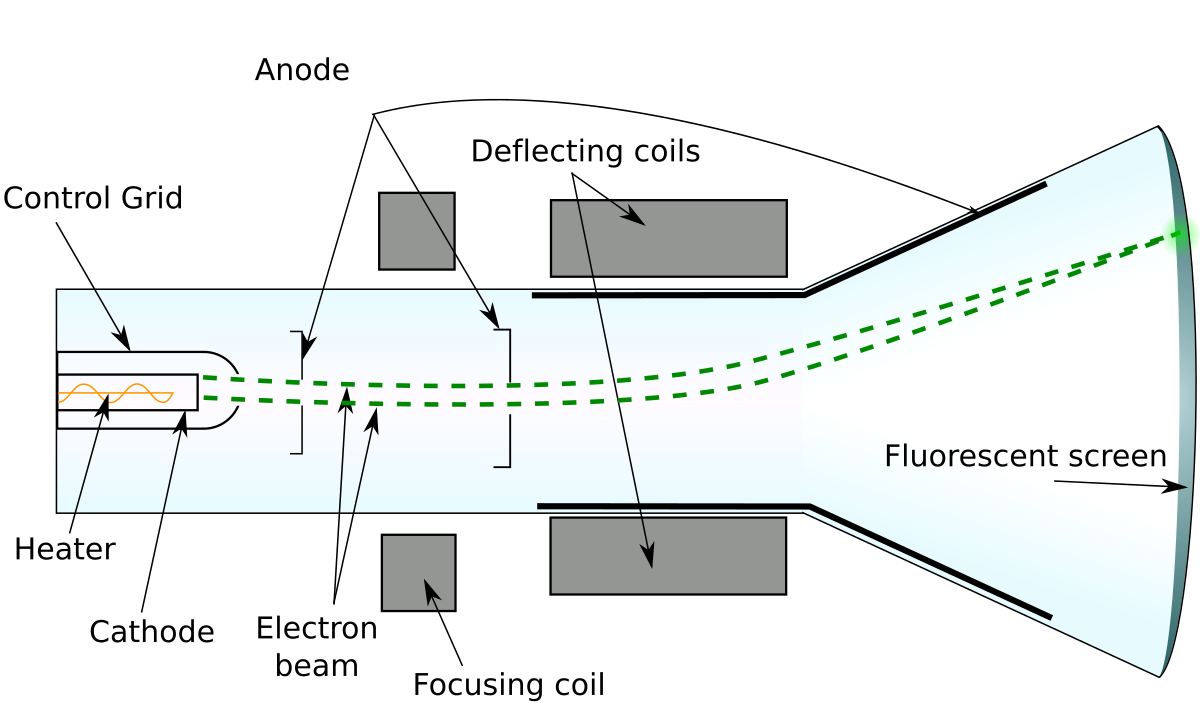
File Cathode Ray Tube Diagram En Svg Wikimedia Commons
Q Tbn 3aand9gctsnzmvc G1jsalbowayhttut W Qbmsdlrbbgsbgx1g Ibrx87 Usqp Cau
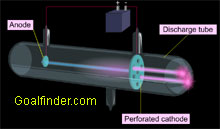
Goalfinder Discovery Of Proton Animated Easy Science Technology Software Online Education Medical K12 Animation E Learning
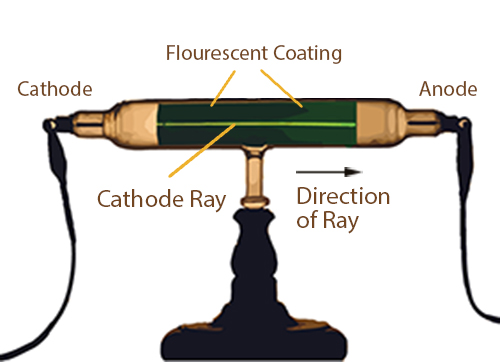
Models Of The Atom Animation Annenberg Learner
Q Tbn 3aand9gcsvckcybrl4 2jvdaa4zabnqj 3yxwi9c8ija Usqp Cau
Cathode Ray Tube Crt Science Facts
Schoolphysics Welcome

Charge To Mass Ratio Electron Class 11 Chemistry Youtube
Ieeexplore Ieee Org Iel7 Pdf
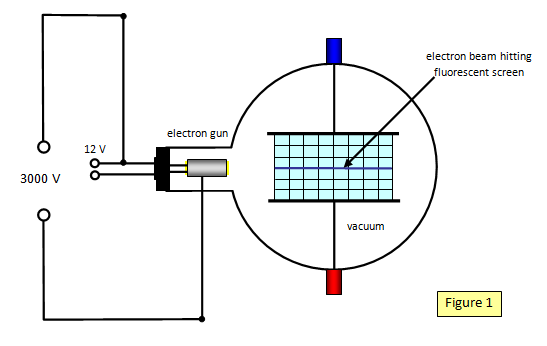
Schoolphysics Welcome

Cathode Ray Wikipedia

Electronic Structure Of Atoms Chemistry Library Science Khan Academy

Gcse Igcse Physics Part1 Cathode Ray Tube From Cro Cathode Ray Oscilloscope Video Youtube

Cathode Rays Introduction To Chemistry

Discharge Tube Experiment And Properties Of Cathode Rays

Ernest Rutherford S Gold Foil Experiment Physics Lab Video Lesson Transcript Study Com

Cathode Ray Discharge Tube Construction Youtube

Production Of X Rays
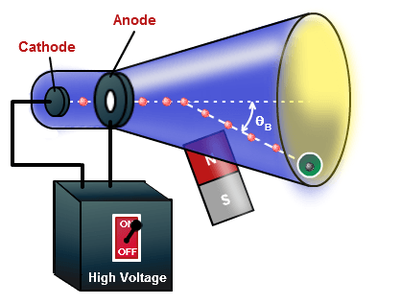
Atomic Timeline Project Sutori

Cathode Ray Tube Youtube

Chemistry 2 Storyboard By 6d7057b5

Click Here If Animations Are Not Working Ppt Download

Signal Generator Connected To A Cathode Ray Oscilloscope Or Cro Stock Photo Alamy

Modern Theories Of The Atom Ppt Download

Q Tbn 3aand9gcs4tjagl1f0ppqzombrcfffvshoxhta Feqeq Usqp Cau

Crookes Tube Wikipedia

Further Understanding Of The Atom Ck 12 Foundation

Cathode Ray Discharge Tube Experiment Experiments Science Electricity Atomic Structure
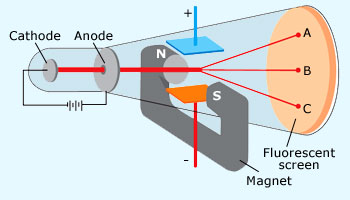
Cathode Ray Experiment

Q Tbn 3aand9gcrmzassioyu7r6emebwdpo8oi8zeokv8 Octa Usqp Cau

J J Thomson S Cathode Ray Tube Crt Definition Experiment Diagram Video Lesson Transcript Study Com
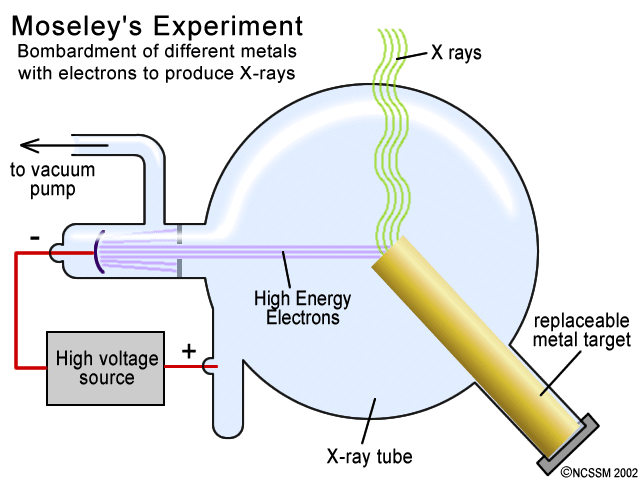
Tiger Ncssm Distance Education And Extended Programs

Learn Electron And It S Discovery In 3 Minutes

Cathode Ray Oscilloscope Cro Electronics Physics Youtube
Cathode Ray Electron Discovery Vacuum Experiment Jean Baptiste Perrin Transparent Png

Cathode Ray Wikipedia
Cathode Ray Tube Technology

J J Thomson S Cathode Ray Tube Crt Definition Experiment Diagram Video Lesson Transcript Study Com
Cathode Ray Tube Definition Charatersitics Diagram Youtube

Media Portfolio

J J Thomson

Atomic Structure

Cathode Ray Experiment Summary Explanation Video Lesson Transcript Study Com
Gcse Igcse Physics Part1 Cathode Ray Oscilloscope Parts From Cro Cathode Ray Oscilloscope Video On Vimeo

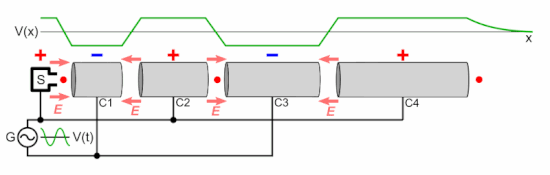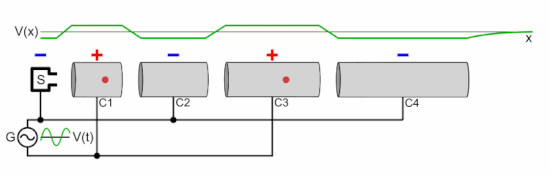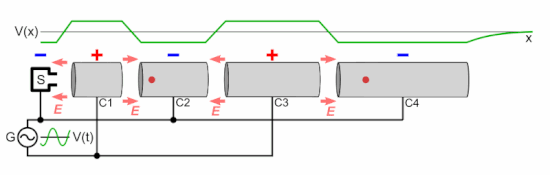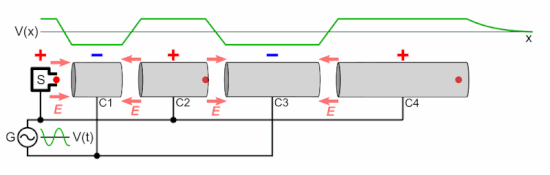
Animations Produced And Used In The Research Download Table

Media Portfolio
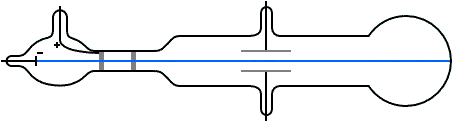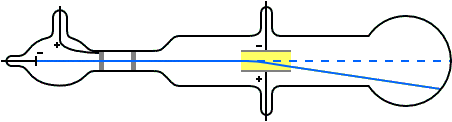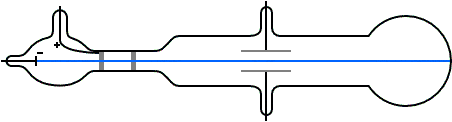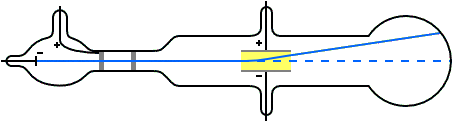
Q Tbn 3aand9gcqg Q366kmi14xzv1z0yrnf Hgnn6 X0nu23q Usqp Cau

Interactive Student Tutorial
Q Tbn 3aand9gcslplcpp4hpif3f0owc7mfxuup1bc O1pqltzqtsqnp7yzkrglq Usqp Cau

3d Animation Cathode Ray Tube 19 Youtube
Go Aws 34vpqhp
Q Tbn 3aand9gcsz8gzafh8bsl5zauw4wlvolydyu8snbidxrg Usqp Cau
Q Tbn 3aand9gcthtcu5v0ohqe2 Fq30y366bkhonsdhch8yofd Hqov6vnpcdvg Usqp Cau

Q Tbn 3aand9gcrdg9thnraixzh5vsuvomwawza9bjgzkxdlhq Usqp Cau

Proton Read Chemistry Ck 12 Foundation
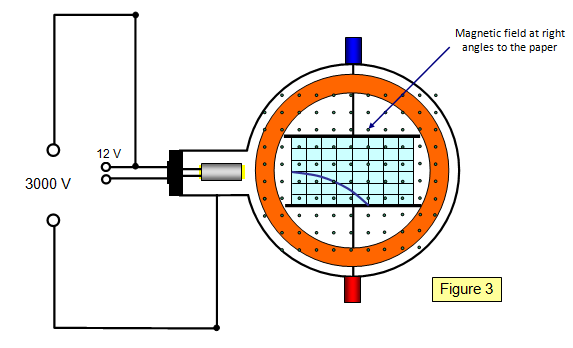
Schoolphysics Welcome

X Rays Storyboard By Valebelieve
Crookes Tube Stem Resource Finder
Q Tbn 3aand9gcqmmemfuczgla97 Pw5852wdkrhjxz9qosbm3a5n Njggp1f6wk Usqp Cau

Q Tbn 3aand9gctzoprxub 25tcyls L1i58snkoklxkguaf0q Usqp Cau
Q Tbn 3aand9gcsqvmicqvowuubf8eiz4vsygpq4autrmbfg0a Usqp Cau

551 Millikan Experiment On A Single Electron Charge Measurement Oil Drop Experiment Experiments Electrons

Thomson S Cathode Ray Tube Experiments Youtube

Q Tbn 3aand9gcrzjoumg Uigncpmqj9nlf8hwgu Kv87fgsvw Usqp Cau
Cathode Ray Oscilloscope And Its Applications On Vimeo
J J Thomson Robert Millikan Atomic Model Project123

Cathode Ray Wikipedia

Eeee Storyboard Av 5e161ec8

Discovery Of Cathode Rays And Anode Rays And J J Thomson Atomic Model Youtube
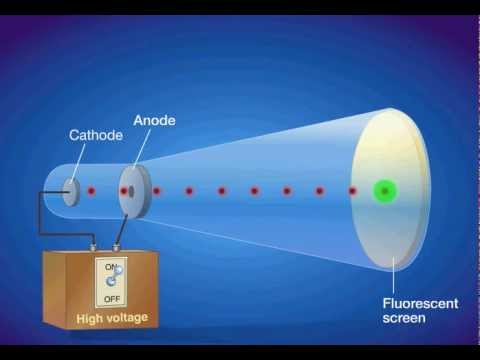
Cathode Ray Oscilloscope

Cathode Ray How Does It Work Animated With 3d Youtube
Cathode Ray Tube Youtube
Chemistry Technology Storybvoard Storyboard By Kailahg
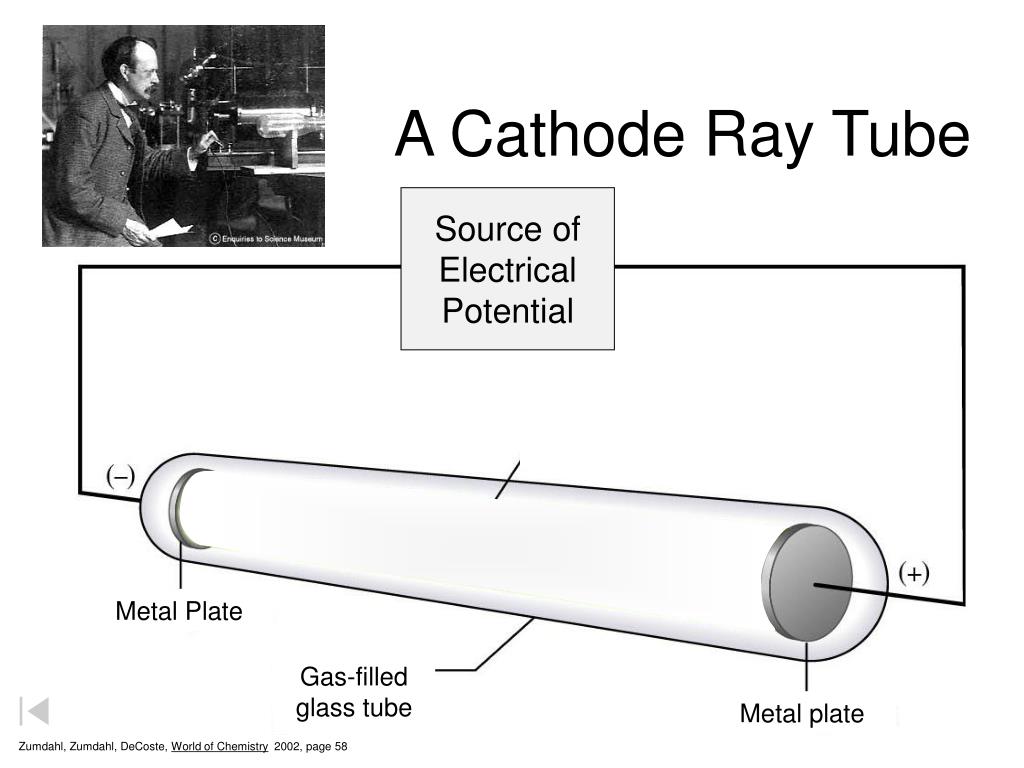
Ppt A Cathode Ray Tube Powerpoint Presentation Free Download Id

J J Thomson S Cathode Ray Tube Crt Definition Experiment Diagram Video Lesson Transcript Study Com

Q Tbn 3aand9gcsx9nhq3n8mt Ziat0q B8w17ppv3s4m Glig Usqp Cau
Q Tbn 3aand9gcssph7sv9kk8r75onswscfxz 6f1otb2syn G Usqp Cau

X Ray Production Animation Youtube

Goalfinder Discovery Of Proton Animated Easy Science Technology Software Online Education Medical K12 Animation E Learning
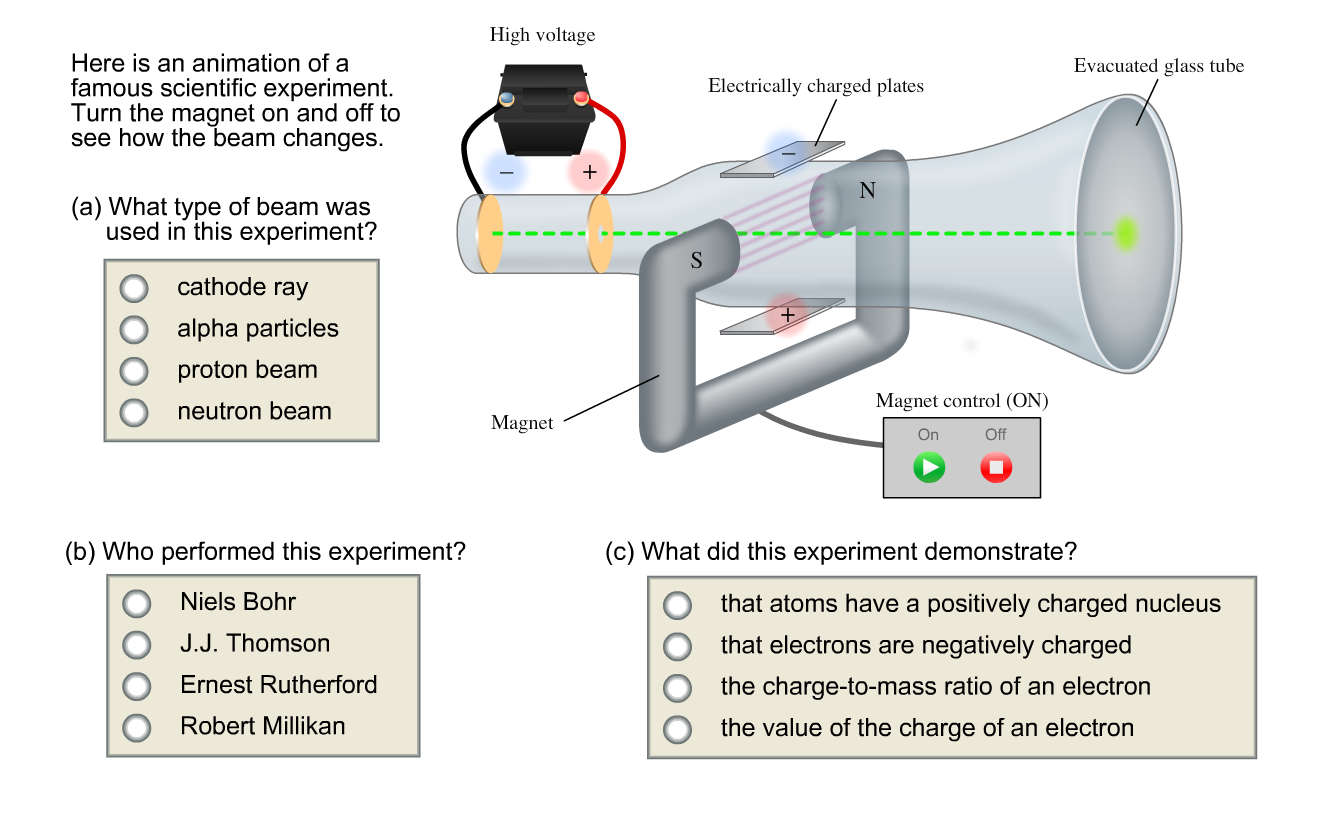
Solved Here Is An Animation Of A Famous Scientific Experi Chegg Com
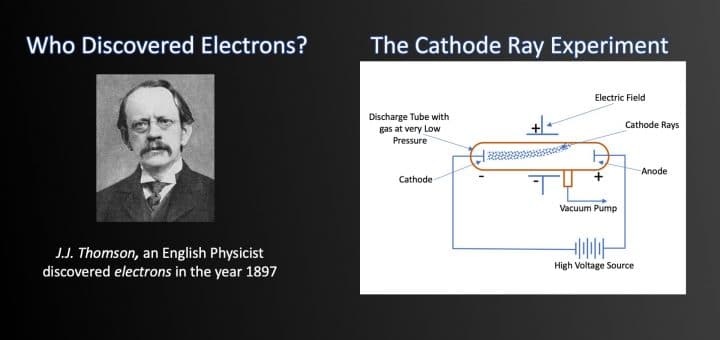
Who Discovered Electrons The Cathode Ray Experiment Selftution

Q Tbn 3aand9gcrlozveqnsexqb9ya54wysplqxzlssxyv9xwq Usqp Cau

Learn Electron And It S Discovery In 3 Minutes

Excellent Explanation Discovery Of The Electron Cathode Ray Tube Experiment This Really Helped My Kids Understa Apologia Chemistry Basic Physics Experiments

Physicslab Famous Discoveries The Franck Hertz Experiment



